
Discover how "inactive" ingredients in generic drugs can cause unexpected reactions and how to identify and manage excipient sensitivities for better health.
Read More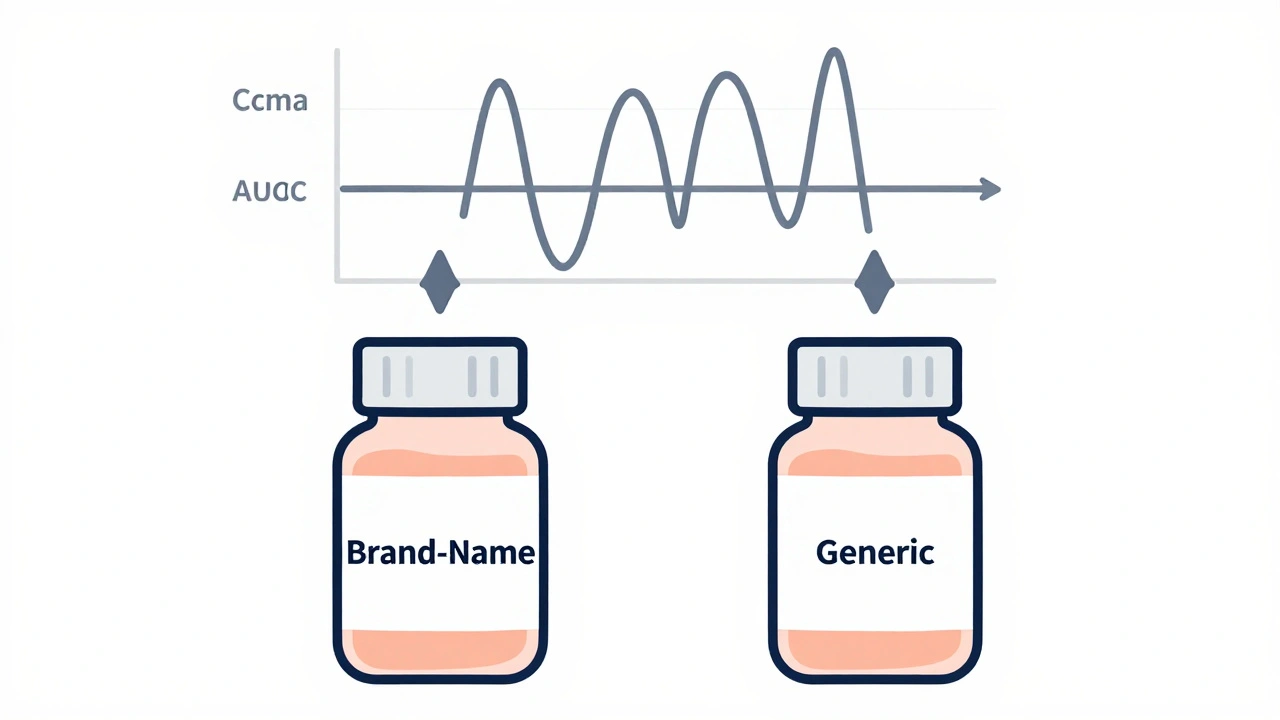
Bioequivalence is the scientific standard the FDA uses to prove generic drugs work the same as brand-name versions. Learn how Cmax and AUC measurements, the 80-125% rule, and clinical trials ensure generics are safe and effective.
Read More