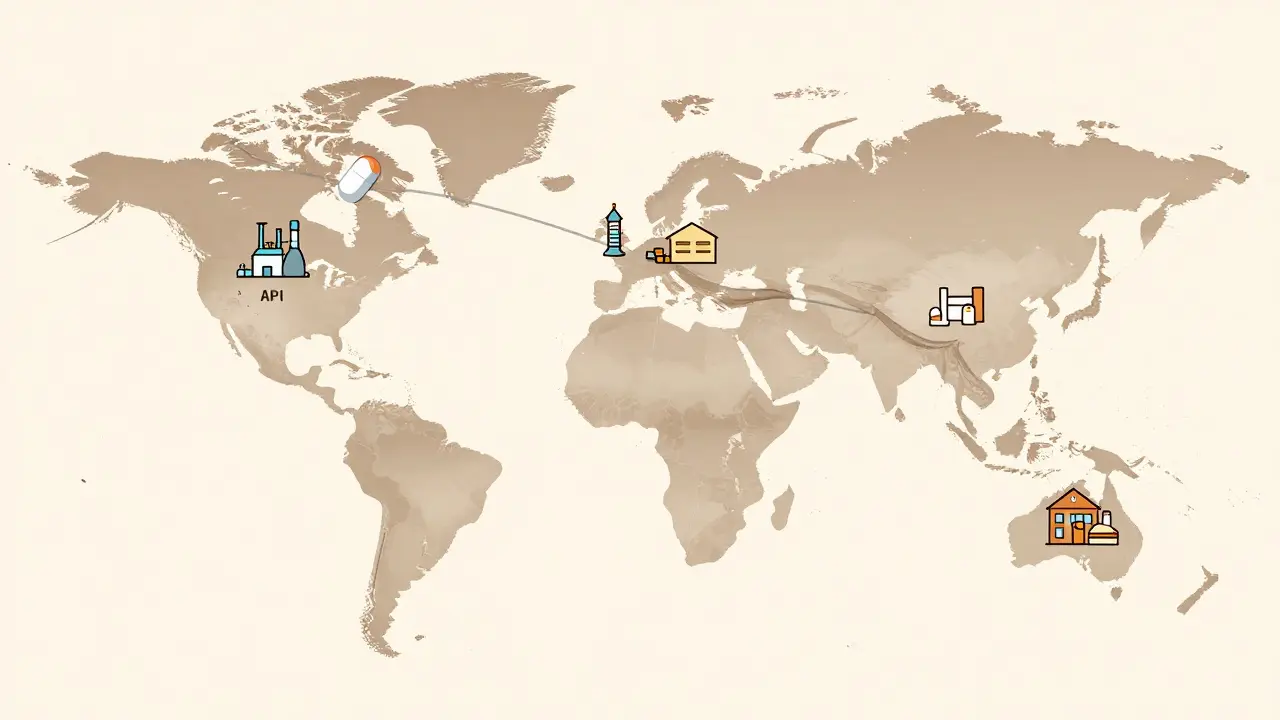
Every time you pick up a prescription for a generic drug-say, the 10 mg tablet of atorvastatin for cholesterol-you’re holding the end result of a long, complicated journey. It doesn’t start in the pharmacy. It doesn’t even start in the factory. It starts thousands of miles away, in a chemical plant in China or India, where the raw ingredient-the Active Pharmaceutical Ingredient (API)-is made. From there, it travels through layers of manufacturers, distributors, and middlemen before landing on your pharmacy shelf. And yet, most people have no idea how it got there. Why does a $5 generic drug sometimes cost $15 at the counter? Why do some medications suddenly disappear for months? The answer lies in the generic drug supply chain.
Where It Begins: The API That Makes the Medicine
Before you ever see a pill, you need the active ingredient. That’s the API. For a generic version of a blood pressure drug, it’s the exact same molecule as the brand-name version. But here’s the twist: less than 12% of global API manufacturing happens in the United States. Over 88% of it happens overseas, mostly in China and India. These countries have the infrastructure, cheap labor, and regulatory frameworks to produce massive volumes of APIs at low cost. That’s why generic drugs are affordable in the first place.
But this global setup has risks. During the early days of the pandemic, when lockdowns hit India and China, over 170 generic medications vanished from U.S. shelves. No API meant no pills. The FDA responded by ramping up inspections of foreign facilities-from 248 in 2010 to 641 in 2022. But inspections can’t catch everything. Quality control becomes a gamble when your raw material comes from halfway around the world.
From Ingredient to Pill: Manufacturing and FDA Approval
Once the API is shipped to a drug manufacturer, the real work begins. The manufacturer doesn’t just mix the powder into capsules. They must follow strict Good Manufacturing Practices (GMP), which cover everything from air filtration in the factory to how often equipment is cleaned. Every batch is tested. Every label is checked. One mistake can mean a recall, a fine, or worse-harm to patients.
Before any of this can even happen, the manufacturer has to get approval from the FDA. For generics, that means submitting an Abbreviated New Drug Application (ANDA). The FDA doesn’t require new clinical trials. Instead, they only need proof that the generic works the same as the brand-name drug. This is why generics can hit the market so fast after a patent expires. But approval doesn’t mean easy profits. Generic manufacturers operate on razor-thin margins. In fact, they only keep about 36% of what’s spent on their drugs. The rest? Taken by everyone else in the chain.
The Middlemen: Wholesalers and Distributors
Once pills are made, they don’t go straight to pharmacies. They go to wholesale distributors. Companies like AmerisourceBergen, Cardinal Health, and McKesson buy drugs in bulk from manufacturers. They store them in giant warehouses. Then, they sell them to pharmacies-retail chains like CVS or Walgreens, or small independent shops.
Here’s where pricing gets messy. The price you see on the invoice is called the Wholesale Acquisition Cost (WAC). But that’s not what pharmacies actually pay. Distributors offer discounts-sometimes called prompt payment discounts-to get paid faster. A pharmacy that pays within 10 days might get 5% off. A big chain that buys millions of pills a year might get 15% off. Independent pharmacies? They get less. That’s why big chains can afford to sell generics cheaper than small shops.
And here’s another layer: distributors often carry multiple brands of the same generic drug. One might be made in India. Another in Germany. One might cost $1.20 per pill. Another, $1.80. The pharmacy chooses based on price and availability. But they don’t always know why one version is cheaper. The manufacturer? They rarely talk directly to the pharmacy. The distributor is the middleman-and they take their cut.

The Hidden Player: Pharmacy Benefit Managers (PBMs)
Most people don’t realize it, but the person who actually decides how much a pharmacy gets paid to fill your prescription isn’t your insurance company. It’s a Pharmacy Benefit Manager, or PBM. There are three big ones-CVS Caremark, OptumRX, and Express Scripts-and together, they handle about 80% of all prescriptions in the U.S.
Here’s how it works: PBMs negotiate with manufacturers and wholesalers to get discounts. But here’s the key difference between brand and generic drugs. For brand drugs, PBMs often get rebates-sometimes huge ones-because manufacturers want to be on their formulary. For generics? Almost never. Generic manufacturers don’t have the leverage. They can’t afford to play the rebate game. So PBMs use a different tool: the Maximum Allowable Cost (MAC).
MAC is a cap. It’s the most a pharmacy can be reimbursed for a specific generic drug-say, 10 mg atorvastatin. That cap is usually based on the average price of all versions of that drug on the market. But here’s the problem: if the pharmacy buys the drug for $1.50 per pill and the MAC is $1.30, they lose 20 cents every time they fill it. A 2023 survey by the American Pharmacists Association found that 68% of independent pharmacy owners say MAC pricing is below their actual cost. That’s not a business. That’s a charity.
How Pharmacies Survive
So how do pharmacies stay open when they’re losing money on generics? They don’t. Many have shut down. Others rely on volume. A big pharmacy chain might lose a few cents on each generic pill but make it up on the 200 other prescriptions they fill that day. Independent pharmacies? They’re stuck. Some negotiate directly with distributors for better prices. Some switch to generics made in the U.S. even if they cost more. Others just stop stocking certain drugs altogether.
And then there’s inventory. Pharmacies have to keep stock on hand. But generics are unpredictable. One month, a drug is everywhere. The next, it’s gone. Real-world data from companies like Purple Lab helps pharmacies track shortages before they happen. But most small shops don’t have access to that tech. They just wait for the phone to ring-and hope the next shipment arrives.

Why This System Is So Fragile
The generic drug supply chain was designed to be efficient. It worked. Today, 90% of prescriptions in the U.S. are for generics. And they cost only 23% of what brand drugs do. That’s a win.
But it’s a win built on thin margins, global dependencies, and opaque pricing. The manufacturers are squeezed. The distributors take their cut. The PBMs set the reimbursement cap. The pharmacies get stuck paying more than they’re paid. And the patient? They just want their pill.
There’s no single villain here. But there’s a pattern: everyone in the chain is trying to protect their slice of the pie. And the patient ends up paying the price-not just in dollars, but in access. When a manufacturer can’t make money on a generic, they stop making it. When a pharmacy can’t afford to stock it, they stop carrying it. And when a drug disappears? It’s not because of a shortage. It’s because the system broke.
What’s Changing Now
Some changes are starting to happen. The FDA is pushing to speed up generic approvals. The Inflation Reduction Act is forcing Medicare to negotiate prices on some drugs-though most generics aren’t covered yet. Manufacturers are trying new tactics: using AI to forecast demand, blockchain to track shipments, and diversifying their API sources so they’re not reliant on one country.
But without transparency, none of it matters. Nobody knows how much a drug really costs from farm to pharmacy. No one tracks who’s making what profit at each step. Until that changes, the system will keep breaking-just in different ways.
Next time you fill a generic prescription, remember: that pill traveled across oceans, passed through dozens of hands, and survived a financial gauntlet just to get to you. And the reason it’s still here? Not because the system is perfect. But because someone, somewhere, still believes it’s worth keeping alive.
Why are generic drugs cheaper than brand-name drugs?
Generic drugs are cheaper because they don’t need to repeat expensive clinical trials. They only need to prove they’re the same as the original brand. That cuts development costs dramatically. Plus, multiple manufacturers can produce the same generic, creating competition that drives prices down. But even with all that, manufacturers still only keep about 36% of what’s spent on generics. The rest goes to distributors, PBMs, and pharmacies.
Where do most active ingredients for generic drugs come from?
Over 88% of Active Pharmaceutical Ingredients (APIs) for generic drugs are made outside the U.S., primarily in China and India. These countries have the infrastructure to produce large volumes at low cost. But this reliance creates vulnerability-supply chain disruptions in those regions can cause nationwide drug shortages.
Why do pharmacies sometimes lose money on generic drugs?
Pharmacies often lose money because they’re reimbursed based on the Maximum Allowable Cost (MAC), which is set by Pharmacy Benefit Managers (PBMs). The MAC is usually lower than what the pharmacy paid to buy the drug from distributors. If a pharmacy buys a pill for $1.50 but gets paid only $1.30 by insurance, they lose 20 cents per prescription. This is especially common for independent pharmacies.
What role do Pharmacy Benefit Managers (PBMs) play in the generic drug supply chain?
PBMs act as middlemen between insurers, pharmacies, and drug manufacturers. They negotiate discounts, create formularies, and set reimbursement rates. For generics, they use Maximum Allowable Cost (MAC) to cap how much pharmacies get paid. Unlike with brand drugs, PBMs rarely get rebates from generic manufacturers because those companies can’t afford to offer them. This gives PBMs outsized control over what pharmacies earn.
Are generic drugs safe if they’re made overseas?
Yes. The FDA requires all generic drugs, regardless of where they’re made, to meet the same safety and quality standards as brand-name drugs. That includes inspections of foreign manufacturing sites. The FDA increased inspections of overseas facilities from 248 in 2010 to 641 in 2022. While supply chain risks exist-like delays or quality lapses-there’s no evidence that overseas-made generics are less safe than those made in the U.S.
Why do some generic drugs suddenly disappear from shelves?
Generic drugs disappear because manufacturers stop making them. This usually happens when the profit margin is too thin. If a manufacturer can’t make money on a drug after paying for ingredients, labor, shipping, and regulatory compliance, they’ll quit. That’s why shortages often hit older, low-cost generics first. The system rewards volume, not variety.
Is there a way to make the generic drug supply chain more reliable?
Yes. Experts suggest three main fixes: diversifying API sources to avoid over-reliance on one country, using technology like AI and blockchain to track supply chain delays, and reforming reimbursement models so pharmacies aren’t forced to sell drugs at a loss. Some states are already experimenting with price transparency laws. But real change will require coordinated action from regulators, manufacturers, PBMs, and pharmacies.





Amadi Kenneth
March 17, 2026 AT 10:24Shameer Ahammad
March 17, 2026 AT 13:18Alexander Pitt
March 17, 2026 AT 23:36Manish Singh
March 18, 2026 AT 19:15Nilesh Khedekar
March 18, 2026 AT 23:01Laura Gabel
March 20, 2026 AT 16:16Melissa Starks
March 22, 2026 AT 15:32Kal Lambert
March 24, 2026 AT 02:33Melissa Stansbury
March 24, 2026 AT 04:51cara s
March 25, 2026 AT 00:53Robin Hall
March 26, 2026 AT 07:45Suchi G.
March 26, 2026 AT 16:08Andrew Muchmore
March 26, 2026 AT 17:47